Axolotl My Time Has Come To Burn I Invoke The Ancient Power That I May Return, A-X-O-L-O-T-L my time has come to burn i invoke the ancient power that i may return, 1.08 MB, 00:47, 441,539, Jesus the Gamer1000, 2021-04-10T00:01:36.000000Z, 19, MY TIME HAS COME TO BURN by 20percentcooldash on DeviantArt, 20percentcooldash.deviantart.com, 862 x 927, png, burn come deviantart, 20, axolotl-my-time-has-come-to-burn-i-invoke-the-ancient-power-that-i-may-return, KAMPION
Correct option is b) maximum no. Of electrons in 4f orbitals =14; Maximum number of electron that may be present on 4forbital is a 2 b 4 c 7 d 14. To determine the maximum number of electrons present in the 4f orbital, first we need to find out the azimuthal quantum number.
Experts are tested by chegg as specialists in their subject area. We review their content and use your feedback to keep the quality high. View the full answer. The f sublevel as a whole can hold up to 14 electrons due to the fact that it consists of 7 orbitals, but each one can only hold up to 2 electrons. One may also ask, how many sets of quantum number values are there for a 4f electron? 4) each orbital described above (the 4, 2, 2 and 4, 3, 2 values) can hold two electrons each. So, this is the answer to the question: The maximum number of electrons that can have the quantum numbers n = 4 and m ℓ = 2 is four. It shows the electrons in numbers, it doesn’t show the details on the spin of electrons like the orbital diagram.
Csonn t2 atomic structure
PPT - S-orbitals PowerPoint Presentation, free download - ID:1830175
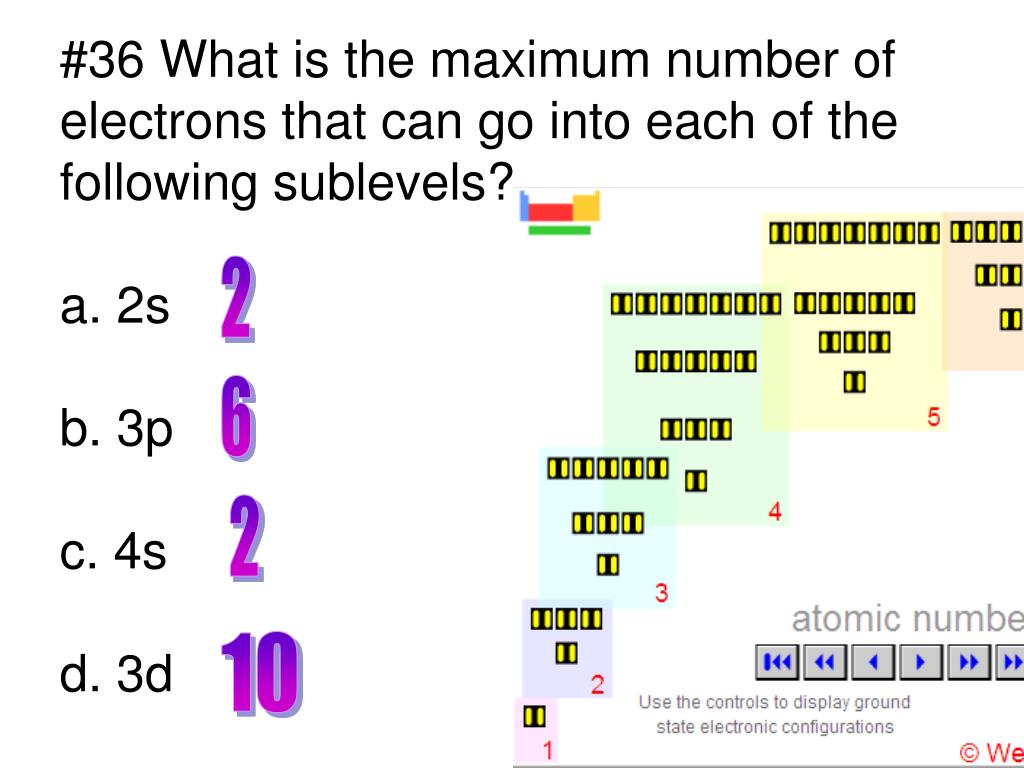
EFellows28 My Chemistry: March 2016

Electron config
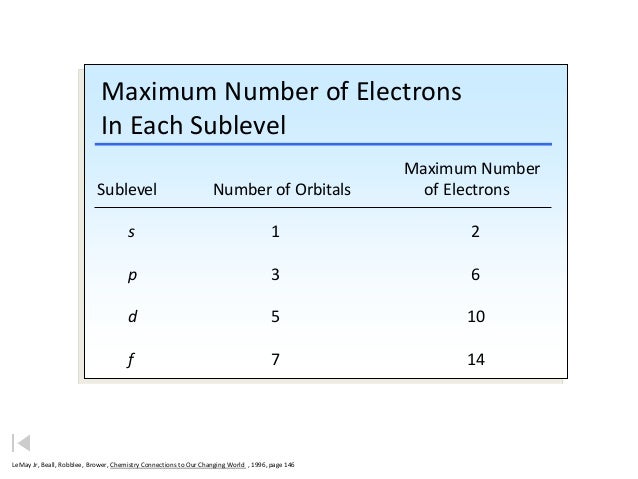
Mec chapter 2

Quantum Number | Periodic Table | Chemogenesis
What are the maximum number of electrons that can be accommodated in 4d
CH150: Chapter 2 – Atoms and Periodic Table – Chemistry
PPT - Electron Configurations and Orbital Diagrams PowerPoint
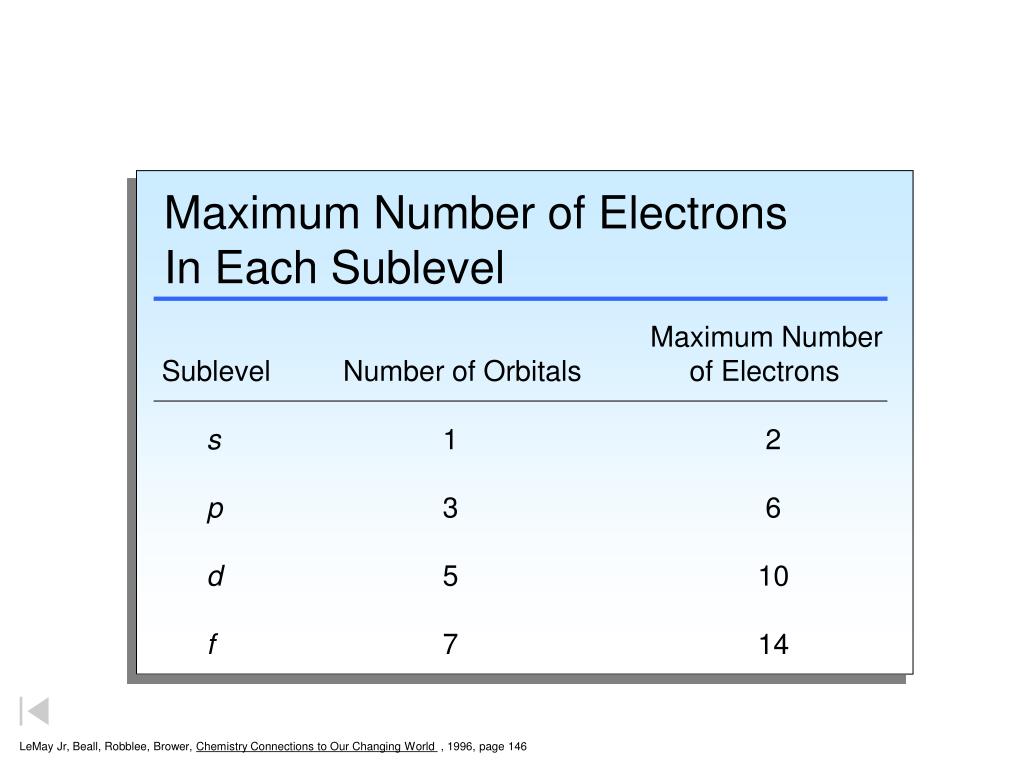
Quantum Numbers, Orbitals, and Probability Patterns | CK-12 Foundation

EmoticonEmoticon